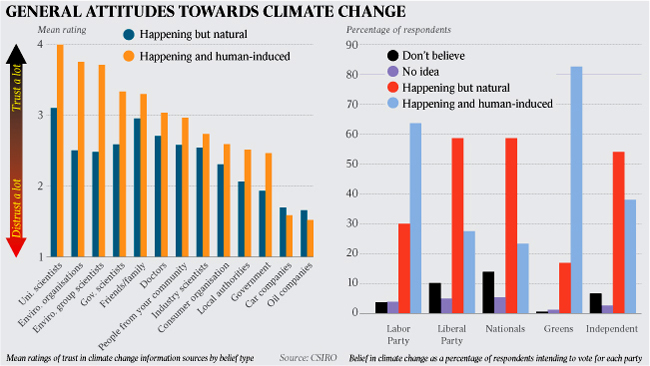
Quotation TMQ1









| Â |
*BL Units |
|
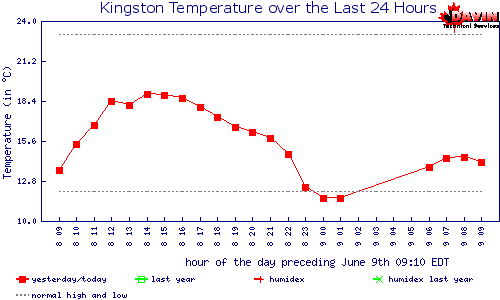
Energy Earth receives from Sun, per day |
1,000,000 |
|
Energy generated within the Earth, per day |
213 |
|
Total energy used by Man, per day |
77 |
|
Energy generated from fossil fuels, per day |
62 |
|
Energy radiated off by Earth, per day |
1,000,275 |
|
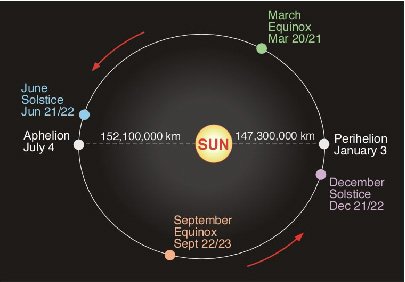
Extra energy received from Sun on January 3 compared to July 4 |
70,000 |
|
Energy received from Sun when Earth when most distant, per day |
965,000 |
|
Energy used immediately to evaporate water, per day |
250,000+ |
|
Energy needed to heat all the CO2 in the atmosphere 1 deg C |
139 |
|
Energy moved north by the Gulf Stream, per day |
7,840 |
|
Energy needed to melt 1000 square km of sea ice 1 metre thick |
22 |
|
Energy generated by the largest hydrogen bomb ever built by the US |
5 |
|
Energy released in a tropical hurricane, per day |
2 |
*1 BL Unit is equal to 4.28400 x 109 kWh (kilo-Watt-hours) or 1.54224 x 1016 joules (1.54224 x 1013 kJ).
 .
.  .
.